A szinaptikus fiziológiáról szóló új tanulmányunk a PNAS-ben már olvasható!
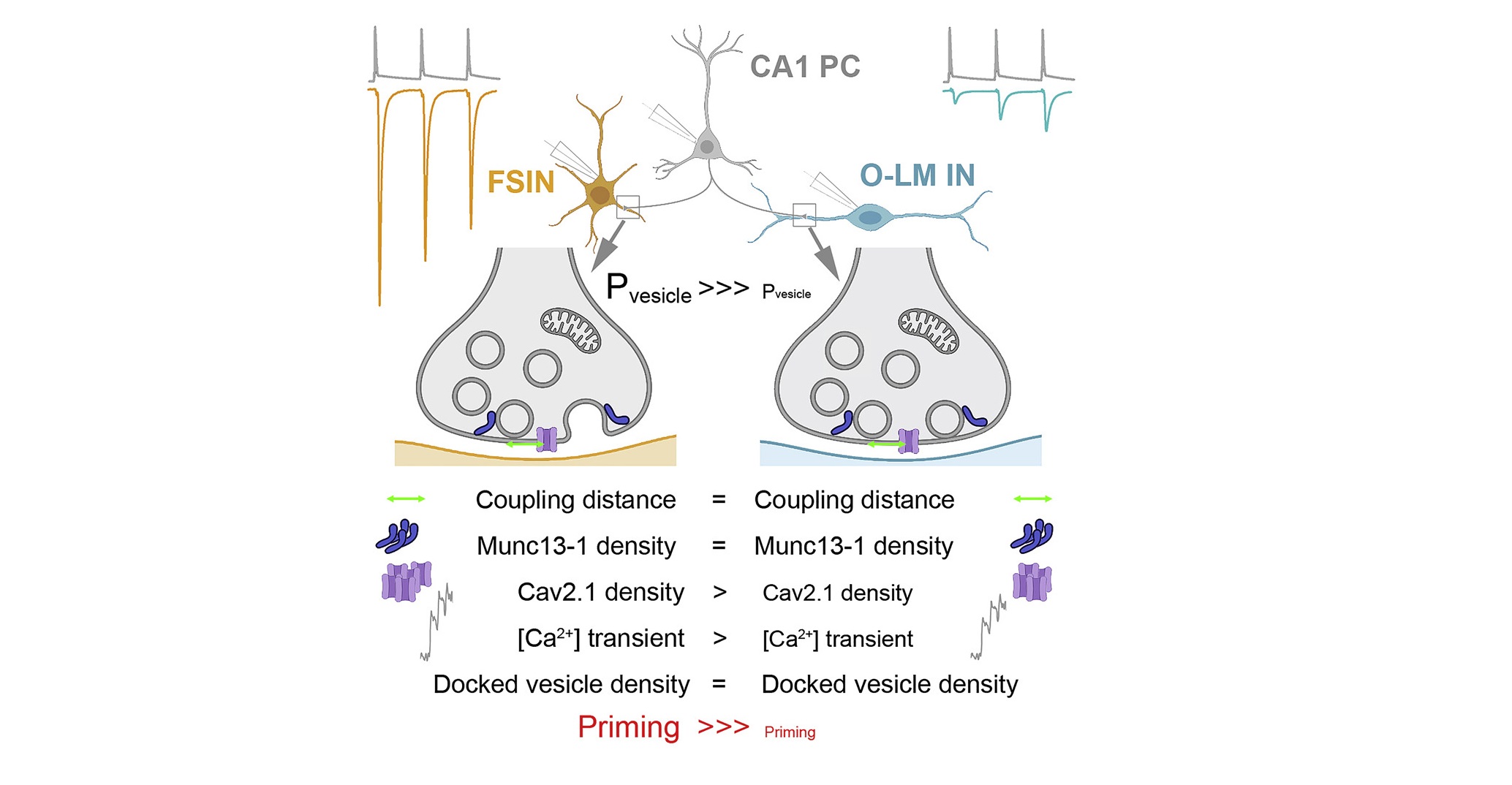
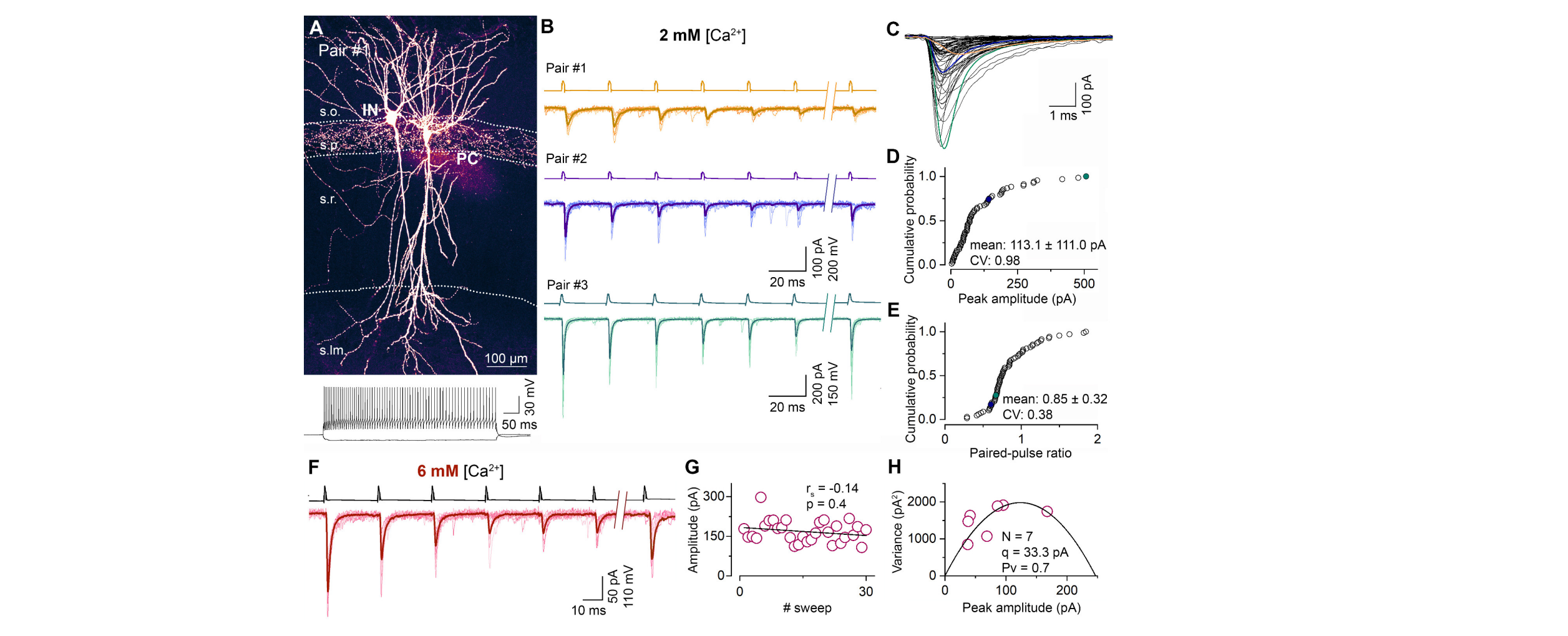
A szinaptikus diverzitás a neuronhálózatok egyik legfontosabb jellemzője. A sokféleség stabilizálja a hálózati aktivitást és növeli a számítási kapacitást. A szinaptikus diverzitás egyik legérdekesebb példája a preszinaptikus felszabadulási valószínűség (Pv) és a rövid távú plaszticitás függése a posztszinaptikus célsejt típusától. Ugyanazon axon két, csak néhány mikrométerrel elválasztott terminálisa különböző Pv-vel jellemezhető, akár nagyságrendi különbség is lehet a glutamát felszabadulás valószínűségénben, a posztszinaptikus célsejtek típusától függően. Ezt korábban azzal a különbséggel magyarázták, hogy milyen valószínűséggel szabadulnak fel a membrán-dokkolt vezikulák egy akciós potenciál hatására. Jelen munkában teszteljük és megerősítjük azt a hipotézist, hogy a sokféleséghez való jelentősebb hozzájárulás a vezikuláris primingben rejlik.