Varietes delectat or the diversity of synapses
Using a combination of state-of-the-art physiological and anatomical methods, Zoltán Nusser's group has succeeded in discovering how an axon of the same nerve cell can form synapses of different strengths with two different nerve cells. Short answer: synaptic vesicles are in different maturation states in weak and strong synapses. Their work was published in Neuron.
For a change, all I found on the Internet is the Hungarian translation of Cicero's "varietas delectat", based on Euripides' comment, as a Hungarian translation: variety delights. Unfortunately, I have nothing new to say to anyone, although how nice it would have been to write a short and light historical introduction to Zoltán Nusser's Neuron article on the reasons for the diversity of synapses!
I didn't give up to find something that might spark the interest of those of us who have the unusual problem of how our unique computer, our brain, which we carry with us and in us, works at the level of neuronal connectivity and synapses. I was a little unsure. To what extent are diversity and variety synonymous, exact equivalents of each other at the neural level? But when it came to diversity, I was confronted with the Greek Euripides, who lived in the fifth century BC, and Stephen, our first king, who was called a saint, who was born four centuries later than Cicero, who was, any case, more familiar and more authoritative to us. He considered diversity so important that he reminded his son of its importance in a work that still survives today. "For as guests come from different lands and provinces, so they bring with them different languages and customs, different examples and weapons, and all this adorns the country and makes the court shine. ...a country of one language and one custom is weak and frail."
And we well know that there is no other organ in the whole world with so many cells, so intricately constructed, as the brain - the realm of the mind.
Not only is diversity not easy to study, but sometimes it is not even easy to explain the results. The not-so-long-established practice of graphically summarising the results before the article is a great help. A good graph is a great help even if you have to ask questions.
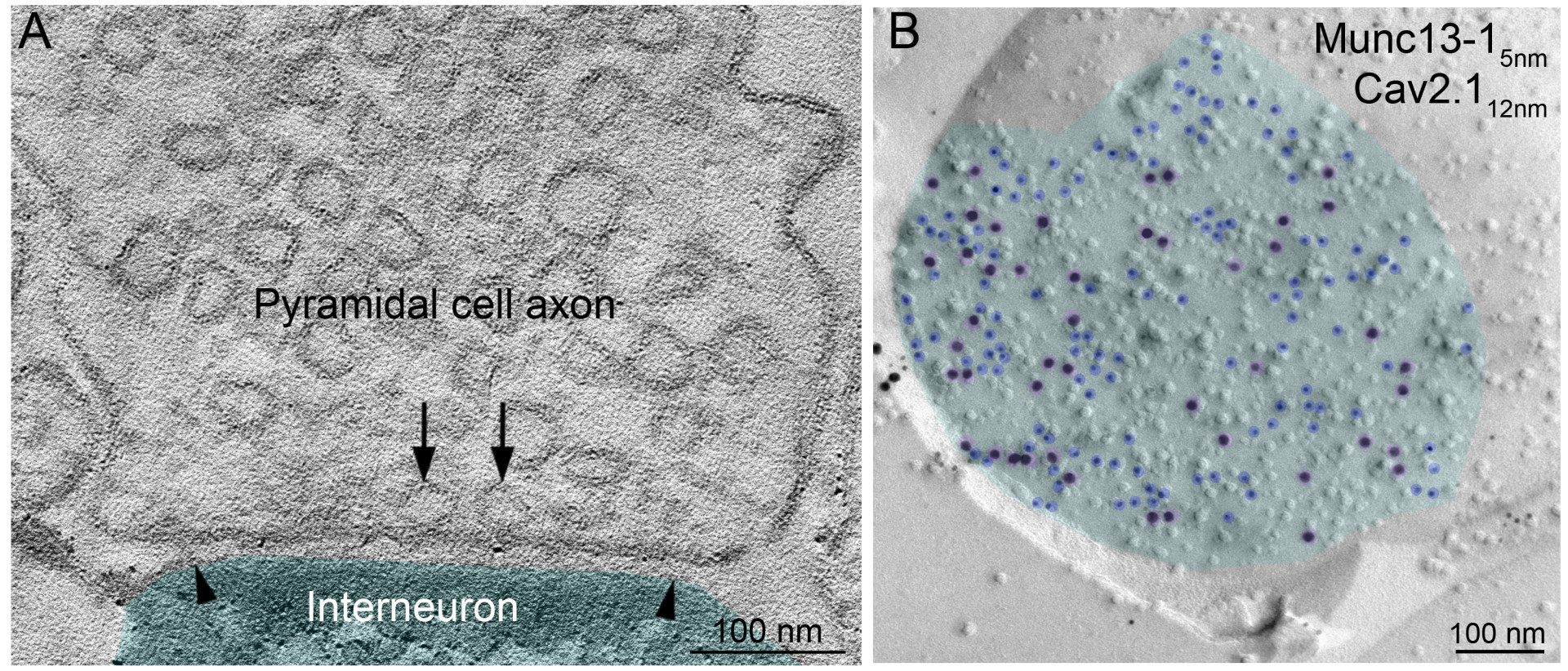
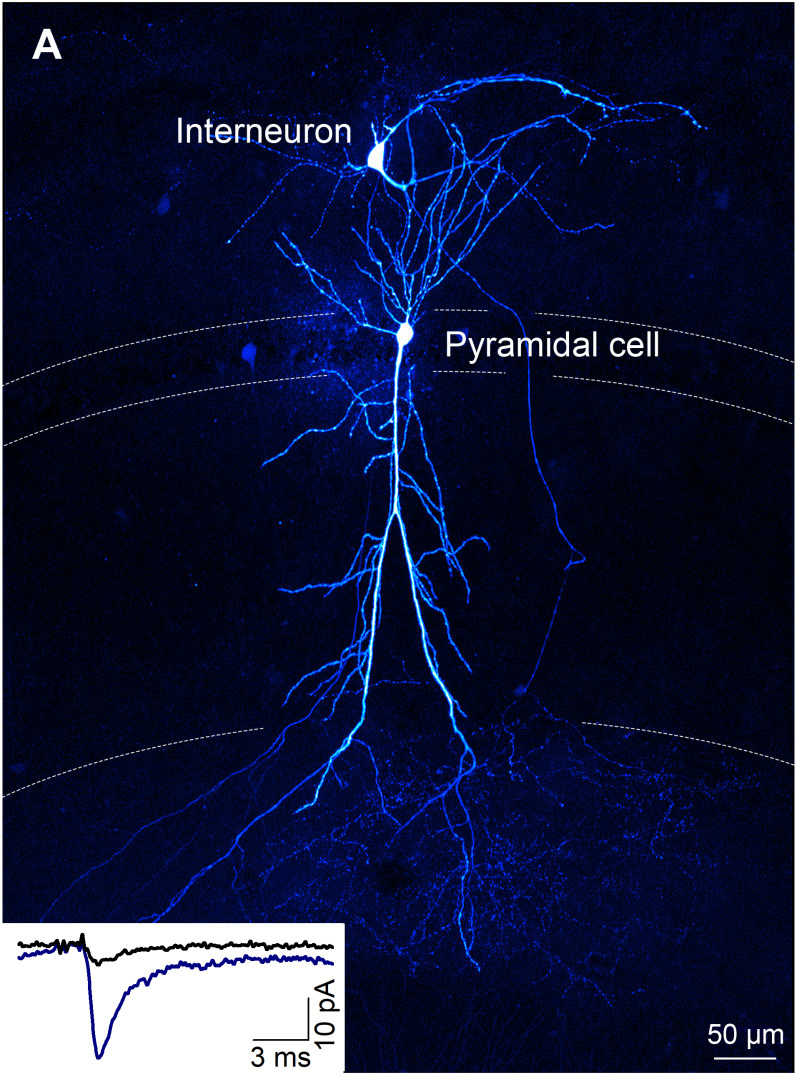
- Nerve cells can be connected by chemical and electrical synapses. You have studied chemical synapses, in which the axon terminal (terminal or bouton) contains vesicles (vesicles) containing nerve signalling substances. When these vesicles fuse with the membrane of the nerve terminal, they are stimulated to release their contents into the synaptic cleft, which then act on receptors on the other nerve cell. You have been looking for an explanation of how an axon of the same nerve cell can form synapses of different strengths with two different nerve cells.
Why is this an important question?
Zoltán Nusser
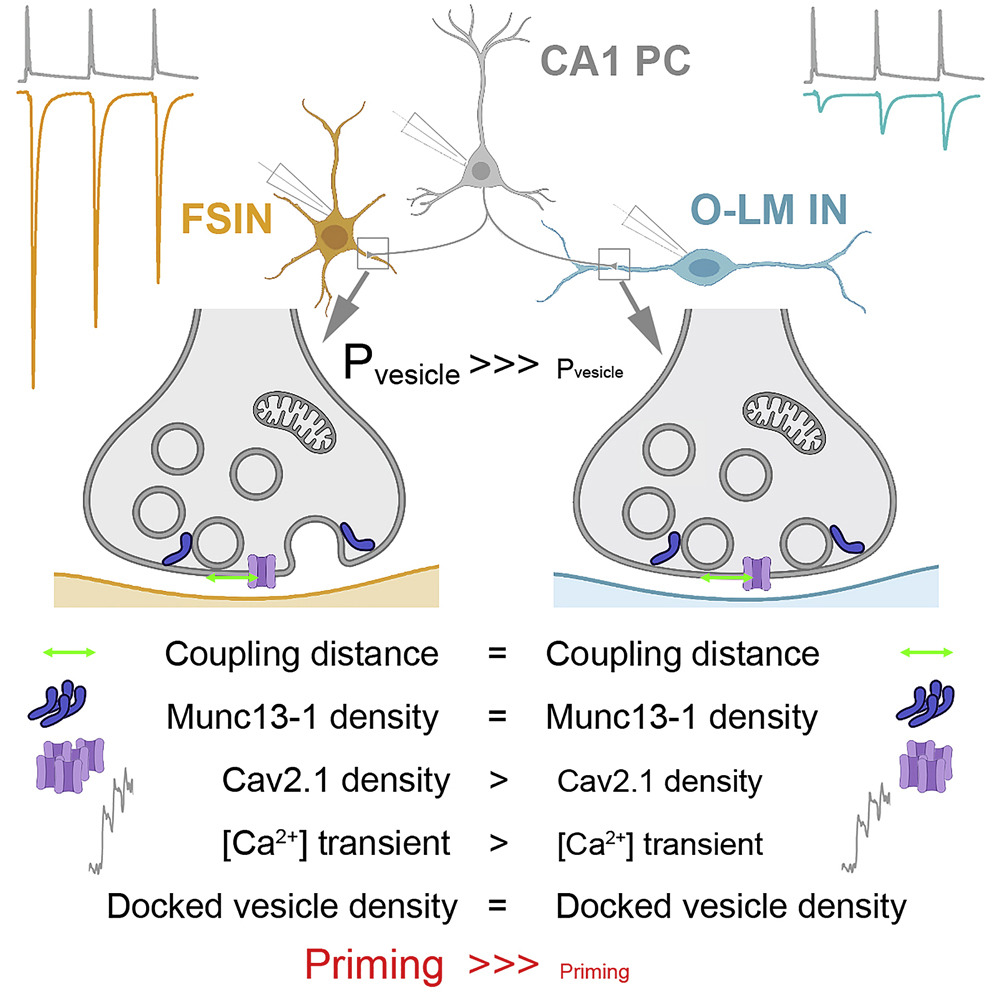
- The function of neuronal networks is determined by the different efficiencies of the chemical synapses that connect neurons. It has been known for decades that synapses of the same type can also be structurally and functionally different. Although it has been the focus of intense research in recent decades, only some details of the molecular processes underlying synaptic diversity have been revealed. An example of synaptic diversity is when an axon of the same neuron forms synapses of different strengths depending on the type of target cell.
- Starting from what stage of development of the nervous system is synaptic diversity essential?
NZ
- I do not know of any nervous system in which all synapses are of equal strength.
Such postsynaptic target site-dependent differences have been described in leeches.
- I assume that both weak and strong synapses are required for the formation of healthy/healthy neural networks!
NZ
- I agree to the max. That is what I am assuming.
Andrea Lőrincz (one of the first authors of this article)
- I definitely see the diversity caused by the variation in synaptic strength as an advantage, as it can provide a basis for adaptation, and plasticity, for our brain function to be able to respond flexibly to physiological or even pathological changes in activity. However, it is difficult to say exactly how the network effect of the diversity of stimulatory synapses is exerted since neural networks have different levels of complexity. Network models can help to understand this, but for such a model to give a realistic prediction, it needs to be based on as many experimentally determined parameters as possible. This is why it is important to understand the parameters that determine synaptic strength as precisely as possible. This is perhaps where we have now made a small contribution.
- Your results have revealed yet another process underlying synaptic diversity that may influence the dynamics of neuronal networks, in your succinct formulation "the temporally structured set of action potentials of a population of neurons".
How is it possible to distinguish between "variability-invariance" and abnormal synaptic function in such a complex system?
NZ
- An abnormal (pathological) brain is likely to have the same synaptic diversity, only e.g. the whole population is shifted to the right or left (stronger or weaker). Alternatively, the majority of synapses are the same in a patient as in a healthy one, but the weight of specific synapses (e.g. between a pyramidal cell (PC) and a specific interneuron (IN)) varies.
- In one of your experiments, you were able to alter synaptic strength by pharmacological modification. Did only the pyramidal cell tested to have an effect?
NZ
- We think that all drugs have an effect on all terminals. Of course, we show precisely that the effect is not the same for all synapses. This is either because the target molecule is different or it is regulated differently.
Noémi Holderith, another first author of the article
- In all the pharmacology experiments we used, we administered the drug in solution, so it acted at all sites where the target molecule was present. Obviously not necessarily with the same potency.
- Your electron tomography and frozen-fracture immunoelectron microscopy images also show how the synaptic vesicles are located in the terminal (also called the terminal apparatus). What determines which synaptic vesicles will be closest to the presynaptic membrane?
NZ
- Unknown, but the simplest explanation is free diffusion. This causes vesicles to come and go within the terminal, which one is closest to the presynaptic membrane is random!
- Is what you observed and described true only for PC cells and only for that type of inhibitory cell, or is it a general phenomenon, true for other brain regions?
NZ
- We believe it is also true for other cortical regions. More specifically, other subregions of the neocortex and hippocampus. There may be other synapses that act in a similar way to the synapses that go to the high-efficiency fast-firing cells, but I think that the particularly low-efficiency synapse that goes to the O-LM cells is probably unique to the cortical network. But until we have data on that, it's just a hunch.
LA
- Yes, our observation is probably true for the connections between neocortical and hippocampal pyramidal cells. However, we are cautious about generalizing to other brain areas. It is precisely in the cerebellum that we observed in a previous collaboration that the spatial distribution of voltage-dependent calcium channels and synaptic vesicles attached to the presynaptic membrane (docked) may differ in weak and strong synapses. This is why we were surprised that pyramidal cells choose a different strategy; in their connections, this different spatial arrangement does not hold.
It should be added, however, that in the cerebellum we did not study the connections of one type of presynaptic cells, but of different types.
- Why did you focus on the CA1 region of the hippocampus? Is it because it is easily accessible or is there another reason?
NZ
As you said, it is easily accessible. But not in vitro but in vivo approaches are relevant for this. If our long-term goal is to study these cells and connections in the brain of a behaving animal, then it makes sense to do our in vitro experiments already in that area, which we can then easily study in the brain of a behaving animal. And that, I think, is the CA1 area of the dorsal hippocampus in addition to the neocortex.
LA
- We also like CA1 from a practical point of view. Its input-specific stratification makes it easy to sample. It is easier to sample large numbers of elements from each synapse type, at least for anatomical studies. For replicate experiments, for example, it was easier to concentrate synapses going to O-LM cells in a part of the stratum oriens rather than scattered.
HN
- The same practical aspect of CA1 is also present in tomograms, where, even just for replica, a random sample of stimulatory inputs is taken. The only difference is that the target cell is identified by in vitro drainage (and filling).
- You have mentioned various experimental procedures, which everyone at the institute knows require a lot of practice and manual dexterity in addition to theoretical knowledge. Who did what type of experiment, what was the biggest challenge for the four first authors, was there a need for any method development?
LA
- There were four shared first authors, which is not so common, but it also suggests that this was a real team effort, with equally important contributions.
Our talented Ph.D. student Mohammad Aldahabi performed physiological and pharmacological experiments on the pyramidal cell - O-LM cell pairs. He is now on a well-deserved holiday in Jordan with his family, but before leaving he successfully presented his results at the KOKI days. Flóra Bálint performed the same measurements on the relationships between pyramidal cells and parvalbumin-expressing interneurons. Flóra is at home having a baby, but fortunately, we were able to celebrate the success of the paper with her recently. Noémi did all the calcium measurements in the pyramidal cell axons and the tomographic analysis of the docked vesicles. And I studied the intrasynaptic (within synapse) distribution of Munc13-1 molecules, which indicate the location of calcium channels and docked vesicles, on freeze-fractured replicas.
Zoli wrote the manuscript and kept everything handy and together as usual.
- You have an enviable technical arsenal and methodological skills at your disposal, but there is always room for improvement and modification. Was there a need for that now?
LA
- Although there was no need for method development this time, we worked with the existing technical repertoire, but I think everyone still had their own challenges. For me personally, it was related to image analysis. For the previous analysis of the distribution of gold particles, we used a program written in Python within the lab. This time, however, without any prior knowledge, I had to work with MATLAB code provided by Maria Reva, who is also the author of the paper. Luckily, I managed to not only use the codes over time but also adapt them to our own questions!
HN
- I should add that some of the in vitro pairwise drains required the so-called "perforated patch" technique, which, although known, is a very difficult technique to implement. It requires great skill and was first implemented in our lab by Mohammad. An additional challenge was to combine [Ca2+] transient measurements with pharmacology because the boutons on the axon are particularly sensitive to islet 2-photon laser measurements (scans) and are very easy to destroy.
Andi and I were also given a common challenge in the review process. We were asked to prove that the fixation (treatment of the tissue with a chemical reagent) and embedding used for tomography preserves the native (natural) distribution of synaptic boutons. We were able to answer this question by combining tomographic preparation with high-pressure freezing, which has been shown to preserve the native structure. Based on the results obtained, we concluded that the immersion fixation procedure we used also preserves the native distribution of synaptic vesicles.
- The question does not require much imagination, but the answer does. How will you continue your studies, and what experiments will follow?
LA
- Mohammad and Zoli will continue to dissect the background of the differences observed in priming in the relationships described in this article in an exciting collaboration with Nobel Prize winner Erwin Neher.
We'll stay tuned for other synapses. We would like to better understand the intrasynaptic arrangement of additional key pre- and postsynaptic molecules with nanometric precision. For this, we will mainly use the super-resolution STED microscopy technique.
We also plan to extend our studies to healthy and pathological human tissue.
HN
- Since the article did not explore the hitherto unknown molecular mechanism regulating the differences in the maturation process of vesicles, I will continue in this direction.