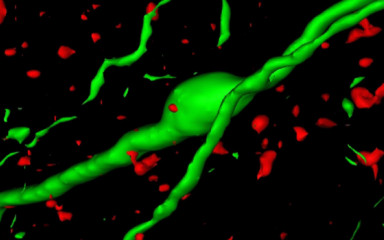
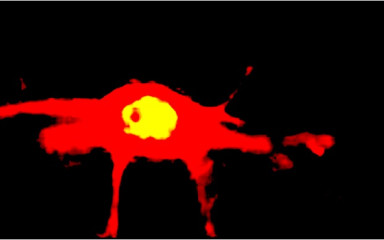
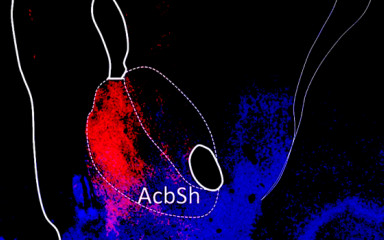
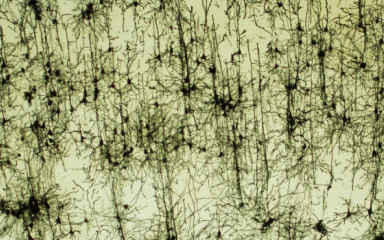
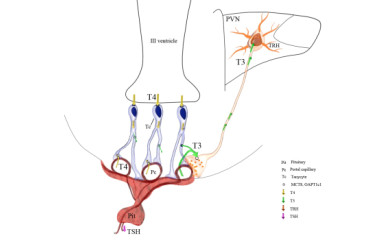
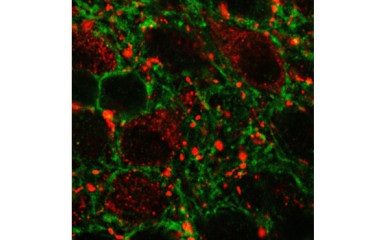
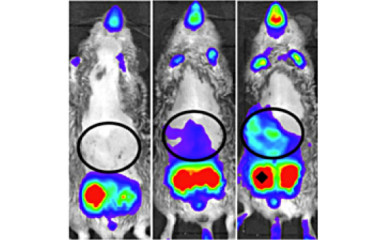
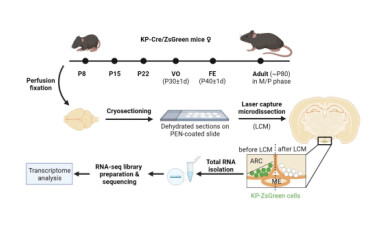
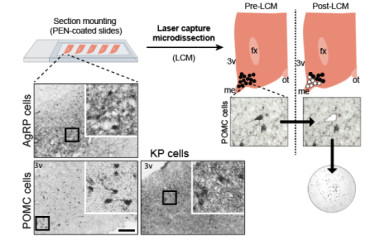
Neuronal control of endocrin activity
Endocrine systems coordinate the functioning of different organs, energy balance and reproduction in cooperation with the central nervous system. Disruption of the coordination of these processes results in severe pathological conditions of the body involving several organ systems. Our institutional research strategy is to understand the central nervous system mechanisms that regulate energy balance, linking stress and metabolic regulation, and may provide the basis for the development of new personalised therapeutic strategies.
(longer)
In Europe, obesity has reached epidemic levels, leading to serious and extremely costly health problems, reducing life expectancy, e.g. type 2 diabetes, cardiovascular disease. Despite these serious health and socioeconomic impacts, we do not have effective anti-obesity drug therapies. Therefore, the exploration of mechanisms regulating energy balance is of paramount importance, which may contribute to provide new anti-obesity drug targets for the pharmaceutical industry.
Similarly, the biological basis of reproductive decline, which is also to a large extent linked to endocrine disruption, is a major problem. Genetic disorders, metabolic disturbances, stress effects, the ageing process and artificial compounds that influence endocrine functions can disrupt the hypothalamic neuronal network responsible for reproduction, causing disturbances of central nervous system origin as well as infertility.
The institute's research strategy is to focus on the neuroscience of endocrine disruption, combining state-of-the-art genomics, metabolomics, transgenics and imaging techniques to elucidate the neuroscience processes that may lead to infertility and morbid obesity.